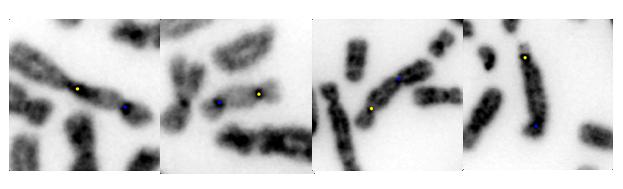
Dicentric chromosomes detected with Cytognomix’s proprietary method. Centromeres are labeled with yellow and blue dots.
Ionizing radiation produces characteristic chromosome changes. The altered chromosomes contain two central constrictions, termed centromeres, instead of one (known as dicentric chromosomes [DCs]). Chromosome biodosimetry is approved by the IAEA for occupational radiation exposure, radiation emergencies, or monitoring long term exposures. In emergency responses to a range of doses, labs need efficient methods that identify DCs.
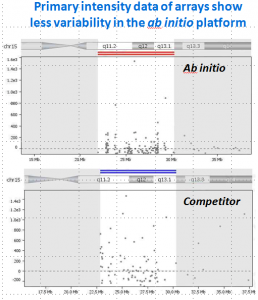
Cytognomix has developed a novel approach to find DCs that is independent of chromosome length, shape and structure from different laboratories (paper: TBME). The Automated Dicentric Chromosome Identifier and Dose Estimator (ADCI) software examines images produced by any of the existing automated metaphase capture systems found in most cytogenetic laboratories.
ADCI is now available for for trial or purchase (link)
Or contact us for details (pricing)
ADCI uses machine learning based algorithms with high sensitivity and specificity that distinguish monocentric and dicentric chromosomes (Try the Dicentric Chromosome Identifier web app). With novel image segmentation, ADCI has become a fully functional cytogenetic biodosimetry system. ADCI takes images from all types of commercial metaphase scanning systems, selects high quality cells for analysis, identifies dicentric chromosomes (removing false positives), builds biodosimetry calibration curves, and estimates exposures. ADCI fulfills the criteria established by the IAEA for accurate triage biodosimetry of a sample in less than an hour. The accuracy is comparable to an experienced cytogeneticist. Check out our online user manual: wiki.
Early development of ADCI was supported by the US Public Health Service 5U01AI091173-02. Product testing is currently supported by the Build-in-Canada Innovation Progam. Working with IBM Canada and Western University, we have developed a high performance computing version, ADCI-BG/Q has demonstrated adequate throughput to handle the workload from a simulated mass casualty event (1000 samples in 1.4 hr; Li et al. IEEE Intnl Conf on Automation for Sustainability, 2012. Pp. 30 – 35 DOI 10.1109/ICIAFS.2012.6420039 (paper: ICIAFS); and paper: Rogan et al. Radiat Prot Dosimetry, 2014).
Some presentations and articles about ADCI:
Yanxin Li, Joan H. Knoll, Ruth Wilkins, Farrah Flegal, and Peter K. Rogan. Automated Discrimination of Dicentric and Monocentric Chromosomes by Machine Learning-based Image Processing. EPR Biodosimetry International conference. Dartmouth College Oct 4-8, 2015. Now published in Microscopy Research and Technique (link). Manuscript (pdf): Li et al 2016. Companion on-line software, the Dicentric Chromosome Analyzer is at: cytobiodose.cytognomix.com
Peter K Rogan, Yanxin Li, Ruth Wilkins, Farrah Flegal, Joan H. Knoll. Radiation dose estimation by automated chromosome biodosimetry. EPR Biodosimetry International conference. Dartmouth College Oct 4-8, 2015. Now published in Radiation Protection Dosimetry (link). Manuscript (pdf): Rogan et al. 2016.